Read
From Our Care Team
Clinical resources, and health & wellness advice

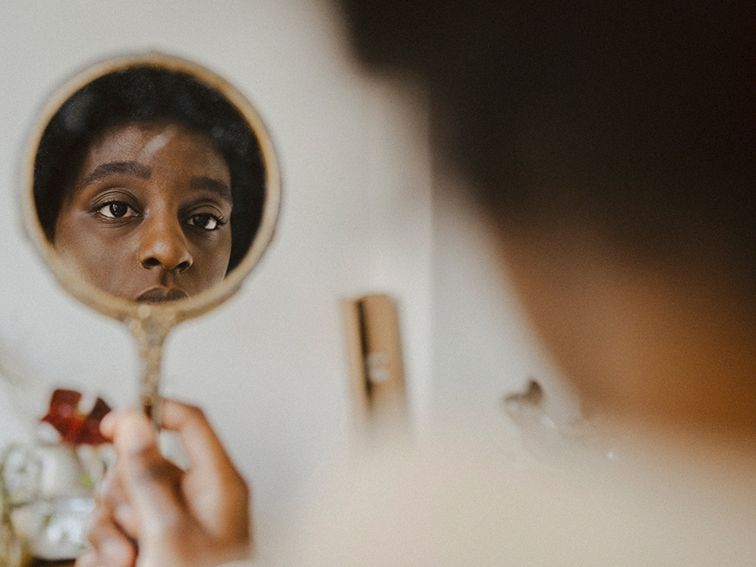
Featured
I'm not stressed. Am I? Oh no.
When you have a lot of stress in your life, it can start to feel normal. You may not even notice your clenched jaw or racing mind. To help you reset, see what Firefly providers had to say about stress in their own lives.

January 1, 2022
Anxiety Disorders: More Than Feeling Anxious

January 1, 2022
Look Out for Added Sugar

March 23, 2022
Celebrating a World of Flavors

September 2, 2021
How Much Alcohol is Safe?

July 14, 2021
The Firefly Hypertension Experience

April 1, 2024
The Many Health Benefits of Fiber

March 22, 2023
Colorectal Cancer Q&A
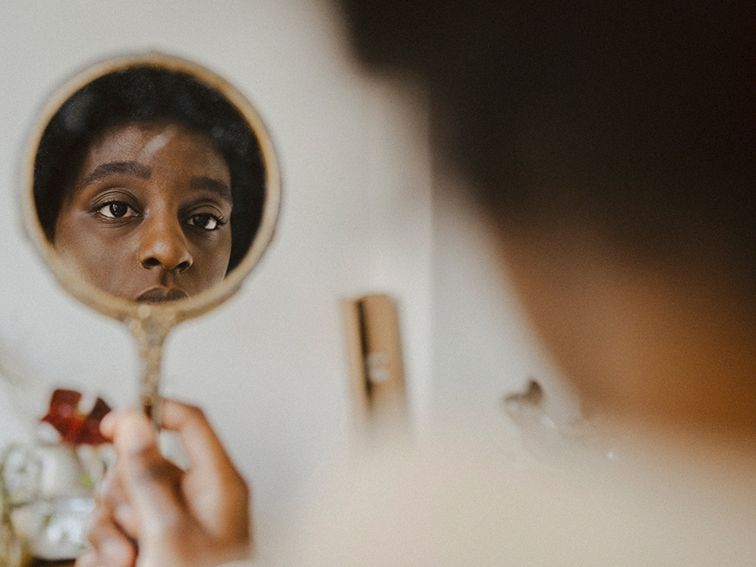
April 1, 2024
I'm not stressed. Am I? Oh no.

September 1, 2021
5 Most Common Dermatology Concerns

January 1, 2022
Cervical Cancer - A Preventable Disease

October 7, 2021
5 Motivational Tips to Help You Reach Your Goals

February 21, 2022
Racial and Ethnic Disparities in Blood Pressure Treatment for African Americans